













中国农业科学 ›› 2021, Vol. 54 ›› Issue (10): 2039-2052.doi: 10.3864/j.issn.0578-1752.2021.10.001
任志杰( ),李倩,孙钰佳,孔冬冬,刘良玉,侯聪聪(
),李倩,孙钰佳,孔冬冬,刘良玉,侯聪聪( ),李乐攻(
),李乐攻( )
)
收稿日期:2021-03-21
接受日期:2021-04-14
出版日期:2021-05-16
发布日期:2021-05-24
联系方式:
任志杰,E-mail: renzj0424@163.com。
基金资助:
REN ZhiJie( ),LI Qian,SUN YuJia,KONG DongDong,LIU LiangYu,HOU CongCong(
),LI Qian,SUN YuJia,KONG DongDong,LIU LiangYu,HOU CongCong( ),LI LeGong(
),LI LeGong( )
)
Received:2021-03-21
Accepted:2021-04-14
Published:2021-05-16
Online:2021-05-24
摘要:
【目的】水稻花期偶遇干热风/干旱,导致脆弱的生殖细胞快速失水,极大地降低产量,这一过程中钙离子作为通用的第二信使传导了干旱或其他逆境信号,但背后的分子机制尚不清楚。分析钙离子透过性胁迫反应阳离子通道家族(calcium-permeable stress-responsive cation channels,CSCs)基因的生理和分子功能,为研究作物干热风的感应机制提供新的理论基础和思路。【方法】采用电生理学和遗传学方法,利用双电极电压钳技术在水稻中鉴定得到一个具有典型特征的受体类-钙通道蛋白,名为OsCSC11,对其蛋白序列进行生物信息学和进化关系分析。运用qRT-PCR和GUS报告基因活性分析确认OsCSC11的表达模式,在拟南芥原生质体细胞和洋葱表皮细胞中瞬时表达OsCSC11-GFP融合蛋白,验证OsCSC11的亚细胞定位;同时利用CRISPR/Cas9基因编辑技术获得OsCSC11的突变体,并通过细胞学等手段分析突变体表型和相关生理功能。【结果】蛋白序列比对发现,OsCSC11具有CSCs家族成员典型的保守结构域DUF221,但与其他成员序列差异大,存在不同于其他成员的特异结构域(motif)属于独立的亚家族。OsCSC11主要在水稻的花药和叶片中表达,进一步分析发现全长OsCSC11处于静息状态,可被高渗透溶液激活;但是删除N端156氨基酸(TM1-3)之后的OsCSC11ΔTM1-3具有组成型的通道活性,特异选择钙、镁二价阳离子;推测TM1-3是这类通道的受体结构域,感应干热风胁迫,而OsCSC11ΔTM1-3区域负责钙信号产生。OsCSC11和OsCSC11ΔTM1-3均定位在细胞质膜上,与其干热风的受体功能相适应。与野生型相比,功能缺失突变体oscsc11-1和oscsc11-2的雄蕊较小、花药表面蹙皱,整体多呈弯曲状态,花粉含水量较低,败育率高达60%—70%。【结论】OsCSC11是水稻感应短期干热风/干旱刺激、介导钙离子内流,调控花药水分状态和花粉发育的受体类钙通道,可能参与了水稻雄蕊应对干热风的原初感应过程。
任志杰,李倩,孙钰佳,孔冬冬,刘良玉,侯聪聪,李乐攻. 水稻CSC11介导干热风/干旱诱导的钙信号调控雄蕊发育[J]. 中国农业科学, 2021, 54(10): 2039-2052.
REN ZhiJie,LI Qian,SUN YuJia,KONG DongDong,LIU LiangYu,HOU CongCong,LI LeGong. OsCSC11 Mediates Dry-Hot Wind/Drought-Induced Ca2+ Signal to Regulate Stamen Development in Rice[J]. Scientia Agricultura Sinica, 2021, 54(10): 2039-2052.
表1
本研究所用的引物"
| 引物名称 Primer name | 引物序列 Primer sequence (5′-3′) | 用途 Purpose |
|---|---|---|
| CSC11-BamHⅠ-F | CGGGATCCATGGGGCCGACCGCGCCGCCGCCGGACGCCG | 蛙卵表达载体构建 Vector construction for oocyte expression |
| CSC11-EcoRⅠ-R | GGAATTCTCAGGATTGATACAGGCTCCAATCC | |
| CSC11ΔTM1-3-BamHⅠ-F | CGGGATCCATGGAGGACGCCCTTCGCA | |
| ProCSC11- Hind Ⅲ-F | CCCAAGCTTTATAGAATGGGTCATCATAGCA | p1300-proCSC11-GUS表达载体构建 Construction of of p1300-proCSC11-GUS vector |
| ProCSC11- BamHⅠ-R | CGGGATCCCGCCGGGGGACGGGGACGTGAC | |
| CSC11- EcoRⅠ-F | GGAATTCATGGGGCCGACCGCGCCGCCGCCGGACGCCG | GFP融合表达载体 GFP-CSC11 expression vector |
| CSC11-BamHⅠ-R | CGGGATCCGGATTGATACAGGCTCCAATCC | |
| CSC11ΔTM1-3-EcoRⅠ-F | GGAATTCATGGAGGACGCCCTTCGCA | |
| Target-F | GCGGCGGGGAGCCGGAGGCG | 基因敲除载体 Gene editing |
| Target-R | CGCCTCCGGCTCCCCGCCGC | |
| CSC11-CRI-F | ACCTCGCGTGATCTAGCCCCACC | 靶点检测及测序引物 Target detection and sequencing primer |
| CSC11-CRI-R | GCTTCTCTCAAGCTGGAGCTCC | |
| 11-qRT-F | GGGCATTCCCAAGACGCT | qRT-PCR检测引物 Primers used for qRT-PCR |
| 11-qRT-R | CCAAGAAATCCTGTTCCGCA | |
| OsACTIN1-F | TCCATCTTGGCATCTCTCAG | |
| OsACTIN1-R | GTACCCGCATCAGGCATCTG |
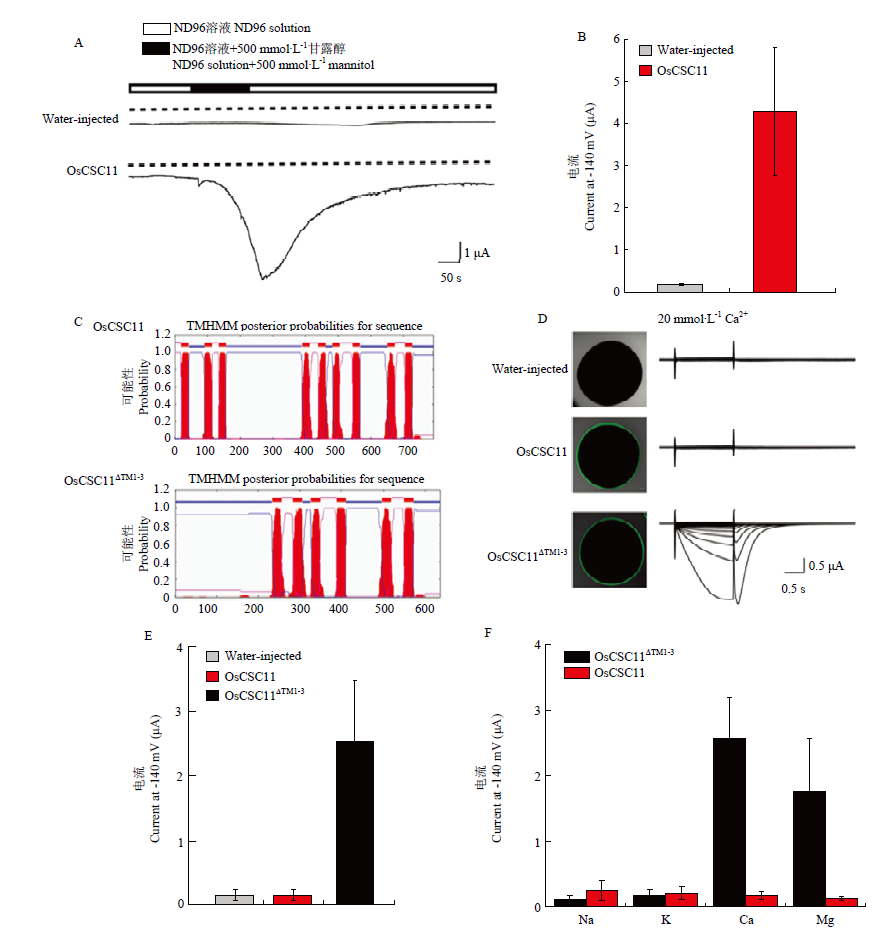
图5
OsCSC11的电生理学分析 A:全细胞记录方式检测表达OsCSC11的蛙卵和对照组注射等体积水的蛙卵在高渗胁迫时产生的电流,电压钳制在-100 mV,横坐标表示记录时间,纵坐标表示电流大小,实线表示电流示踪,虚线表示0 μA电流;B:统计分析A图中记录到的被高渗溶液激活的电流最大值,n>3;C:OsCSC11和OsCSC11ΔTM1-3蛋白的跨膜结构预测;D:OsCSC11和OsCSC11ΔTM1-3蛙卵细胞中的定位以及电生理活性检测,注射水的蛙卵作为对照;E:统计分析图B中钳制电压为-140 mV时的电流最大值,n>5;F:OsCSC11和OsCSC11ΔTM1-3的离子选择性分析,n>5;B、E和F图中的数值由平均值±标准差表示"

| [1] |
SANDERS D, BROWNLEE C, HARPER J F. Communicating with calcium. The Plant Cell, 1999,11:691-706.
doi: 10.1105/tpc.11.4.691 |
| [2] |
GILROY S, TREWAVAS A. Signal processing and transduction in plant cells: The end of the beginning? Nature Reviews Molecular Cell Biology, 2001,2(4):307-314.
doi: 10.1038/35067109 |
| [3] | LEE H J, SEO P J. Ca2+ talyzing initial responses to environmental stresses. Trends in Plant Science, 2021,8:S1360-1385. |
| [4] |
KUDLA J, BECKER D, GRILL E, HEDRICH R, HIPPLER M, KUMMER U, PARNISKE M, ROMEIS T, SCHUMACHER K. Advances and current challenges in calcium signaling. New Phytologist, 2018,218(2):414-431.
doi: 10.1111/nph.14966 |
| [5] |
SWARBRECK S M, COLAÇO R, DAVIES J M. Plant calcium- permeable channels. Plant Physiology, 2013,163(2):514-522.
doi: 10.1104/pp.113.220855 |
| [6] |
HACHEZ C, BESSERER A, CHEVALIER A S, CHAUMONT F. Insights into plant plasma membrane aquaporin trafficking. Trends in Plant Science, 2013,18(6):344-352.
doi: 10.1016/j.tplants.2012.12.003 |
| [7] |
HOU C C, TIAN W, KLEIST T, HE K, GARCIA V, BAI F, HAO Y L, LUAN S, LI L G. DUF221 proteins are a family of osmosensitive calcium-permeable cation channels conserved across eukaryotes. Cell Research, 2014,24(5):632-635.
doi: 10.1038/cr.2014.14 |
| [8] |
ZHANG M, WANG D, KANG Y, WU J X, YAO F, PAN C, YAN Z, SONG C, CHEN L. Structure of the mechanosensitive OSCA channels. Nature Structural & Molecular Biology, 2018,25(9):850-858.
doi: 10.1038/s41594-018-0117-6 |
| [9] |
MURTHY S E, DUBIN A E, WHITWAM T, JOJOA-CRUZ S, CAHALAN S M, MOUSAVI S A R, WARD A B, PATAPOUTIAN A. OSCA/TMEM63 are an evolutionarily conserved family of mechanically activated ion channels. Elife, 2018,7:e41844.
doi: 10.7554/eLife.41844 |
| [10] |
JOJOA-CRUZ S, SAOTOME K, MURTHY S E, TSUI C C A, SANSOM M S, PATAPOUTIAN A, WARD A B. Cryo-EM structure of the mechanically activated ion channel OSCA1.2. Elife, 2018,7:e41845.
doi: 10.7554/eLife.41845 |
| [11] |
LI Q, MONTELL C. Mechanism for food texture preference based on grittiness. Current Biology, 2021,31:1-12.
doi: 10.1016/j.cub.2020.09.070 |
| [12] |
DU H, YE C, WU D, ZANG Y Y, ZHANG L, CHEN C, HE X Y, YANG J J, HU P, XU Z, WAN G, SHI Y S. The cation channel TMEM63B is an osmosensor required for hearing. Cell Report, 2020,31(5):107596.
doi: 10.1016/j.celrep.2020.107596 |
| [13] | LANGRIDGE P, REYNOLDS M. Breeding for drought and heat tolerance in wheat. Theoretical and Applied Genetics, 2021, doi: 10.1007/s00122-021-03795-1. |
| [14] | LAWAS L M F, LI X, ERBAN A, KOPKA J, JAGADISH S V K, ZUTHER E, HINCHA D K. Metabolic responses of rice cultivars with different tolerance to combined drought and heat stress under field conditions. Gigascience, 2019, 8(5): giz050. |
| [15] |
VAN E S S W . Too hot to handle, the adverse effect of heat stress on crop yield. Physiologia Plantarum, 2020,169(4):499-500.
doi: 10.1111/ppl.v169.4 |
| [16] | ZHAO C, LIU B, PIAO S, WANG X, LOBELL D B, HUANG Y, HUANG M, YAO Y, BASSU S, CIAIS P, DURAND J L, ELLIOTT J, EWERT F, JANSSENS I A, LI T, LIN E, LIU Q, MARTRE P, MÜLLER C, PENG S, PEÑUELAS J, RUANE A C, WALLACH D, WANG T, WU D, LIU Z, ZHU Y, ZHU Z, ASSENG S. Temperature increase reduces global yields of major crops in four independent estimates. Proceedings of the National Academy of Sciences of the United States of America, 2017,114(35):9326-9331. |
| [17] |
ZHANG C X, FENG B H, CHEN T T, FU W M, LI H B, LI G Y, JIN Q Y, TAO L X, FU G F. Heat stress-reduced kernel weight in rice at anthesis is associated with impaired source-sink relationship and sugars allocation. Environmental and Experimental Botany, 2018,155:718-733.
doi: 10.1016/j.envexpbot.2018.08.021 |
| [18] |
ARSHAD M S, FAROOQ M, ASCH F, KRISHNA J S V, PRASAD P V V, SIDDIQUE K H M. Thermal stress impacts reproductive development and grain yield in rice. Plant Physiology and Biochemistry, 2017,115:57-72.
doi: 10.1016/j.plaphy.2017.03.011 |
| [19] |
FU G F, SONG J, XIONG J, LIAO X Y, ZHANG X F, WANG X, LE M K, TAO L X. Thermal resistance of common rice maintainer and restorer lines to high temperature during flowering and early grain filling stages. Rice Science, 2012,19:309-314.
doi: 10.1016/S1672-6308(12)60055-9 |
| [20] |
SATAKE T, YOSHIDA S. High temperature-induced sterility in Indica Rices at flowering. Japanese Journal of Crop Science, 1978,47:6-17.
doi: 10.1626/jcs.47.6 |
| [21] | SAKATA T, OSHINO T, MIURA S, TOMABECHI M, TSUNAGA Y. Auxins reverse plant male sterility caused by high temperatures. Proceedings of the National Academy of Sciences of the United States of America, 2010,107:8569-8574. |
| [22] | 曹珍珍. 高温对水稻花器伤害和籽粒品质影响的相关碳氮代谢机理[D]. 杭州: 浙江大学, 2014. |
| CAO Z Z. Mechanism of carbon and nitrogen metabolism related to effects of high temperature on floral organ injury and grain quality of rice[D]. Hangzhou: Zhejiang University, 2014. (in Chinese) | |
| [23] |
MATSUI T, OMASA K, HORIE T. High temperature-induced spikelet sterility of japonica rice at flowering in relation to air temperature, humidity and wind velocity conditions. Japanese Journal of Crop Science, 1997,66(3):449-455.
doi: 10.1626/jcs.66.449 |
| [24] |
SATAKE T, YOSHIDA S. High temperature-induced sterility in indica rice at flowering. Japanese Journal of Crop Science, 2011,47(1):6-17.
doi: 10.1626/jcs.47.6 |
| [25] |
MATSUI T, OMASA K, HORIE T. The difference in sterility due to high temperatures during the flowering period among japonica-rice varieties. Plant Production Science, 2001,4(2):90-93.
doi: 10.1626/pps.4.90 |
| [26] |
ZHAI Y, WEN Z, HAN Y, ZHUO W, WANG F, XI C, LIU J, GAO P, ZHAO H, WANG Y, WANG Y, HAN S. Heterogeneous expression of plasma-membrane-localised OsOSCA1.4 complements osmotic sensing based on hyperosmolality and salt stress in Arabidopsis osca1 mutant. Cell Calcium, 2020,91:102261.
doi: 10.1016/j.ceca.2020.102261 |
| [27] |
LIVAK K J, SCHMITTGEN T D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method . Methods, 2001,25:402-408.
doi: 10.1006/meth.2001.1262 |
| [28] | KOMARI T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant Journal for Cell & Molecular Biology, 2010,6:271-282. |
| [29] |
JEFFERSON R A. The GUS reporter gene system. Nature, 1989,342:837-838.
doi: 10.1038/342837a0 |
| [30] |
YOO S D, CHO Y H, SHEEN J. Arabidopsis mesophyll protoplasts: A versatile cell system for transient gene expression analysis. Nature Protocol, 2007,2:1565-1572.
doi: 10.1038/nprot.2007.199 |
| [31] |
EADY C, WELD R, LISTER C. Agrobacterium tumefaciens- mediated transformation and regeneration of onion (Allium cepa L.). Plant Cell Report, 2000,19:376-381.
doi: 10.1007/s002990050743 |
| [32] |
ZHANG S S, PAN Y J, TIAN W, DONG M Q, ZHU H F, LUAN S, LI L G. Arabidopsis CNGC14 mediates calcium influx required for tip growth in root hairs. Molecular Plant, 2017,10(7):1004-1006.
doi: 10.1016/j.molp.2017.02.007 |
| [33] |
PAN Y J, CHAI X Y, GAO Q F, ZHOU L M, ZHANG S S, LI L G, LUAN S. Dynamic interactions of plant CNGC subunits and calmodulins drive oscillatory Ca2+ channel activities . Developmental Cell, 2019,48(5):710-725.
doi: 10.1016/j.devcel.2018.12.025 |
| [34] |
TIAN W, HOU C C, REN Z J, WANG C, ZHAO F G, DAHLBECK D, HU S, ZHANG L Y, NIU Q, LI L G, STASKAWICZ B J, LUAN S. A calmodulin-gated calcium channel links pathogen patterns to plant immunity. Nature, 2019,572(7767):131-135.
doi: 10.1038/s41586-019-1413-y |
| [35] |
KUDLA J, BATISTIC O, HASHIMOTO K. Calcium signals: The lead currency of plant information processing. The Plant Cell, 2010,22(3):541-563.
doi: 10.1105/tpc.109.072686 |
| [36] |
BERRIDGE M J, LIPP P, BOOTMAN M D. The versatility and universality of calcium signalling. Nature Reviews Molecular Cell Biology, 2000,1:11-21.
doi: 10.1038/35036035 |
| [37] |
DAVENPORT R. Glutamate receptors in plants. Annals of Botany, 2002,90(5):549-557.
doi: 10.1093/aob/mcf228 |
| [38] |
JHA S K, SHARMA M, PANDEY G K. Role of cyclic nucleotide gated channels in stress management in plants. Current Genomics, 2016,17(4):315-329.
doi: 10.2174/1389202917666160331202125 |
| [39] | FORDE B G, ROBERTS M R. Glutamate receptor-like channels in plants: A role as amino acid sensors in plant defence? F1000Prime Reports, 2014,6:37. |
| [40] |
WANG X H, FENG C X, TIAN L L, HOU C C, TIAN W, HU B, ZHANG Q, REN Z J, NIU Q, SONG J L, KONG D D, LIU L Y, HE Y K, MA L G, CHU C C, LUAN S, LI L G. A transceptor-channel complex couples nitrate sensing to calcium signaling in Arabidopsis. Molecular Plant, 2021,14(5):774-786.
doi: 10.1016/j.molp.2021.02.005 |
| [41] |
YUAN F, YANG H, XUE Y, KONG D, YE R, LI C, ZHANG J, THEPRUNGSIRIKUL L, SHRIFT T, KRICHILSKY B, JOHNSON D M, SWIFT G B, HE Y, SIEDOW J N, PEI Z M. OSCA1 mediates osmotic-stress-evoked Ca2+ increases vital for osmosensing in Arabidopsis. Nature, 2014,514(7522):367-371.
doi: 10.1038/nature13593 |
| [42] | DENIS V, CYERT M S. Internal Ca2+ release in yeast is triggered by hypertonic shock and mediated by a TRP channel homologue. Journal of Cell Biology, 2002,156:29-34. |
| [43] |
CATERINA M J, SCHUMACHER M A, TOMINAGA M, ROSEN T A, LEVINE J D, JULIUS D. The capsaicin receptor: A heat- activated ion channel in the pain pathway. Nature, 1997,389(6653):816-824.
doi: 10.1038/39807 |
| [44] |
PEIER A M, MOQRICH A, HERGARDEN A C, REEVE A J, ANDERSSON D A, STORY G M, EARLEY T J, DRAGONI I, MCINTYRE P, BEVAN S, PATAPOUTIAN A. A TRP channel that senses cold stimuli and menthol. Cell, 2002,108(5):705-715.
doi: 10.1016/S0092-8674(02)00652-9 |
| [45] |
MCKEMY D D, NEUHAUSSER W M, JULIUS D. Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature, 2002,416(6876):52-58.
doi: 10.1038/nature719 |
| [46] |
VANNESTE M, SEGAL A, VOETS T, EVERAERTS W. Transient receptor potential channels in sensory mechanisms of the lower urinary tract. Nature Reviews Urology, 2021,18(3):139-159.
doi: 10.1038/s41585-021-00428-6 |
| [47] | ZHOUX L, BATIZA A F, LOUKIN S H, PALMER C P, KUNG C, SAIMI Y. The transient receptor potential channel on the yeast vacuole is mechanosensitive. Proceedings of the National Academy of Sciences of the United States of America, 2003,100(12):7105-7110. |
| [48] |
ZHANG Y F, MARK A H, JAYARAM C, KEN L MUELLER, BOAZ C, WU D Q, CHARLES S ZUKER, NICHOLAS J P R. Coding of sweet, bitter, and umami tastes: Different receptor cells sharing similar signaling pathways. Cell, 2003,112:293-301.
doi: 10.1016/S0092-8674(03)00071-0 |
| [1] | 彭廷燊, 陆久焱, 吴美林, 严雨欣, 刘宏周, 南文斌, 秦小健, 李明, 龚俊义, 梁永书. 多年生水稻黄糯2号和长白7号产量相关性状的QTL分析[J]. 中国农业科学, 2026, 59(7): 1361-1379. |
| [2] | 陈敏, 焦紫岚, 乔承彬, 许昊, 张碧, 马东花, 孔维儒, 王敬文, 宋佳伟, 罗成科, 李培富, 田蕾. 不同亚种水稻种质资源对盐胁迫的形态生理响应及适应策略[J]. 中国农业科学, 2026, 59(4): 705-722. |
| [3] | 郭富城, 唐海江, 郝馨怡, 马国林, 杨九菊, 黄霖锋, 田蕾, 王彬, 罗成科. 不同灌溉方式对宁夏盐渍化土壤水盐运移、水稻产量及水分利用效率的影响[J]. 中国农业科学, 2026, 59(4): 750-764. |
| [4] | 张卫建, 严圣吉, 尚子吟, 唐志伟, 吴柳格, 李佳锐, 陈浩天, 邓艾兴, 张俊, 张鑫, 郑成岩, 宋振伟. 稻田甲烷不完全源于水稻种植的人为贡献[J]. 中国农业科学, 2026, 59(4): 824-833. |
| [5] | 朱姝, 国志鹏, 孙颖. 水稻雷帕霉素靶蛋白OsTOR调控根伸长的功能研究[J]. 中国农业科学, 2026, 59(3): 475-485. |
| [6] | 吕文彦, 程海涛, 马兆惠, 田淑华. 育种新时代水稻杂交育种技术与策略探讨[J]. 中国农业科学, 2026, 59(2): 233-238. |
| [7] | 廖婷璐, 石亚飞, 肖东浩, 舍杨梦斐, 郭富城, 杨九菊, 唐海江, 罗成科. 外源硝普钠对碱胁迫下水稻幼苗糖代谢的影响[J]. 中国农业科学, 2026, 59(2): 265-277. |
| [8] | 刘天生, 刘耕源, 赵安琪, 杨旭, 蔡明雪, 杨艾文, 娄铭轩, 李沐恺, 王晗, 张亚玲. 黑龙江省水稻恶苗病致病群体[J]. 中国农业科学, 2026, 59(2): 305-321. |
| [9] | 王忠妮, 雷月, 李佳丽, 宫彦龙, 朱速松. ABC转运蛋白OsARG1调控水稻抽穗期的功能[J]. 中国农业科学, 2026, 59(1): 1-16. |
| [10] | 费耀莹, 王迪, 唐伟杰, 郭彩丽, 张小虎, 邱小雷, 程涛, 姚霞, 江冲亚, 朱艳, 曹卫星, 郑恒彪. 基于无人机多源影像融合的水稻籽粒蛋白质含量估测[J]. 中国农业科学, 2026, 59(1): 41-56. |
| [11] | 董桂春, 王子涵, 王树深, 李杰, 霍晓晴, 杨瑞, 周娟, 舒小伟, 李妍, 曹靓婧, 王子瑞, 姚友礼, 黄建晔. 硫包衣缓释肥提升水稻产量及氮肥利用率的技术途径[J]. 中国农业科学, 2026, 59(1): 57-77. |
| [12] | 王爱冬, 李瑞杰, 冯向前, 洪卫源, 李子秋, 张晓果, 王丹英, 陈松. 基于多角度成像与机器学习的水稻叶面积精确估算[J]. 中国农业科学, 2025, 58(9): 1719-1734. |
| [13] | 韦萍, 潘炬忠, 朱德平, 邵胜雪, 陈珊珊, 韦雅倩, 高维维. OsDREB1J调控水稻籽粒大小的功能研究[J]. 中国农业科学, 2025, 58(8): 1463-1478. |
| [14] | 刘劲松, 伍龙梅, 包晓哲, 刘志霞, 张彬, 杨陶陶. 短期减施氮肥对华南地区早晚兼用型水稻产量和稻米品质的影响[J]. 中国农业科学, 2025, 58(8): 1508-1520. |
| [15] | 王彬, 吴朋浩, 鲁剑巍, 任涛, 丛日环, 陆志峰, 李小坤. 长江中游地区水稻-油菜轮作体系需水特征[J]. 中国农业科学, 2025, 58(7): 1355-1365. |
|
||