













中国农业科学 ›› 2020, Vol. 53 ›› Issue (9): 1890-1903.doi: 10.3864/j.issn.0578-1752.2020.09.015
白云赫,王文然,董天宇,管乐,宿子文,贾海锋,房经贵,王晨( )
)
收稿日期:2019-09-05
接受日期:2019-12-25
出版日期:2020-05-01
发布日期:2020-05-13
联系方式:
白云赫,E-mail:2017104028@njau.edu.cn。
基金资助:
YunHe BAI,WenRan WANG,TianYu DONG,Le GUAN,ZiWen SU,HaiFeng JIA,JingGui FANG,Chen WANG( )
)
Received:2019-09-05
Accepted:2019-12-25
Published:2020-05-01
Online:2020-05-13
摘要:
【目的】研究vvi-miR160家族(vvi-miR160s)及其靶基因在‘魏可’葡萄种子发育过程中的作用,探究其应答赤霉素(GA)调控葡萄果实无核的潜在机理。【方法】采用miR-RACE、生物信息学分析、RT-qPCR、RLM-RACE等方法,鉴定vvi-miR160家族成员及其靶基因,分析vvi-miR160s及其靶基因应答GA的时空表达模式及其潜在功能。【结果】花前GA处理强烈抑制‘魏可’葡萄胚珠及种子发育,高效诱导其无核,且无核率达99.8%。克隆鉴定了VvMIR160s前体基因序列(501 bp)及成熟体序列,且它们在不同物种间具有较高的保守性;基于vvi-miR160s序列,预测到靶基因VvARF18,利用RLM-RACE技术检测到vvi-miR160s对VvARF18的裂解位点在第10和第11位之间,裂解频度9/17,证明VvARF18是vvi-miR160s的真实靶基因。该靶基因编码683个氨基酸,在398—411位存在核定位信号,且该蛋白亚细胞定位于细胞核上。VvARF18与其他物种间序列的同源保守性较高,基因结构相似,其中VvARF18蛋白与茶、烟草、梅花等物种亲缘关系较近。VvARF18启动子中包含4种作用元件,且含有较多激素相关元件。RT-qPCR结果显示,随着葡萄果实的发育,vvi-miR160c/d/e呈现‘V’形表达趋势,在硬核种子发育期表达较低,而VvARF18表现出与前者相反的表达趋势,在硬核种子发育期呈现高表达,表明vvi-miR160c/d/e对VvARF18负调控,但vvi-miR160a/b与VvARF18表达水平却不存在明显负相关。GA处理极显著地上调了vvi-miR160a/b在葡萄硬核种子发育期的表达,同时也显著抑制了VvARF18在同时期的表达,且它们的表达水平呈负相关,表明GA处理促进了vvi-miR160a/b对VvARF18的负调控作用;相反,GA减弱了vvi-miR160c/d/e对VvARF18的负调控作用。【结论】在vvi-miR160家族中,vvi-miR160c/d/e可能介导VvARF18在葡萄种子发育的特定阶段调控种子的发育形成,而vvi-miR160a/b可能主要介导VvARF18参与调控GA诱导葡萄种子败育的过程。
白云赫,王文然,董天宇,管乐,宿子文,贾海锋,房经贵,王晨. vvi-miR160s介导VvARF18应答赤霉素调控葡萄种子的发育[J]. 中国农业科学, 2020, 53(9): 1890-1903.
YunHe BAI,WenRan WANG,TianYu DONG,Le GUAN,ZiWen SU,HaiFeng JIA,JingGui FANG,Chen WANG. vvi-miR160s in Mediating VvARF18 Response to Gibberellin Regulation of Grape Seed Development[J]. Scientia Agricultura Sinica, 2020, 53(9): 1890-1903.
表1
VvMIR160s PCR扩增引物序列"
| 基因名称 Gene name | 正向引物序列 Forward primer sequence | 反向引物序列 Reverse primer sequence |
|---|---|---|
| VvMIR160a | ACACCTCCTAAAATCATTGTCTG | CTTGTGACATGAATATGGTGCG |
| VvMIR160b | CTATGTATTTGTCTTGTTCTGATTGAA | TGAATGGTCACAGTTCTTTGG |
| VvMIR160c | GGCCTGGCCTCTATAAATATCA | AATCGACCCACAATCAAACC |
| VvMIR160d | GATGTGGTGCTTCGCCAAT | ATGTGGGTTTTCTAAATGCCTAACC |
| VvMIR160e | CACTCACTCACACCCTTCC | ATATTATATTCTCTCTGCAGCCAAG |
表2
vvi-miR160s及VvARF18定量引物序列"
| 基因名称 Gene name | 正向引物序列 Forward primer sequence | 反向引物序列 Reverse primer sequence |
|---|---|---|
| vvi-miR160a | TGACCTTTGTGCTTCAGTGG | GCTATCTGGGTTGACCTCCA |
| vvi-miR160b | TTCTGCAGGAGATGGAGCTT | AGTGTTTCGCCTGCTTGACT |
| vvi-miR160c | CCACATTCCGTGACCTTTCT | GCACAACCCATTTCACCTTT |
| vvi-miR160d | CGCCAATGCAGGAAATTTAT | GGGAGCCAGGCATGTAAGTA |
| vvi-miR160e | CTGTATGCCATTTGCAGAGC | GGGGGAGAAGATTGAAGAGG |
| VvARF18 | CTGAACACGCCTATGGGAAT | CCGTTTCACCCTCAGTGTTT |
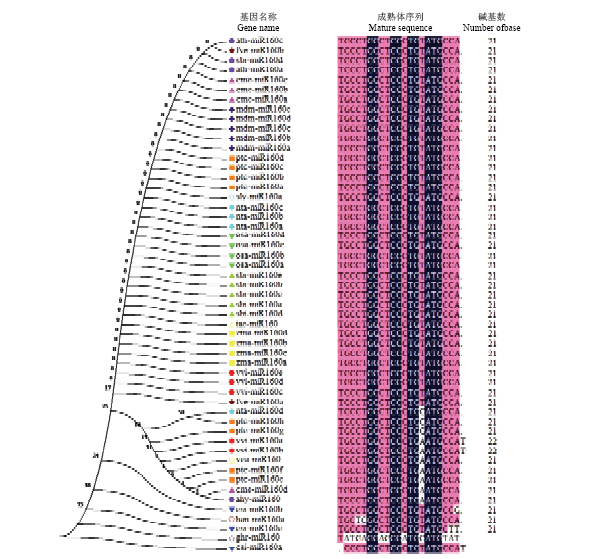
图3
microR160s进化分析和成熟体序列比对 Vv:葡萄Vvtis vinifera;Ha:向日葵Helianthus annuus L;Zm:玉米Zea mays;Ta:小麦Triticum aestivum;Sb:高粱Sorghum bicolor;Os:水稻Oryza sativa;Nt:烟草Nicotiana tabacum;Sl:番茄Solanum lycopersicum;Pt:毛果杨Populus trichocarpa;Cs:柑橘Citurs sinensis;Md:苹果Malus domestica;Gh:棉花Gossypium herbaceum;Cm:甜瓜Cucumis melo;At:拟南芥Arabidopsis thaliana;Ah:花生Arachis hypogaea;Fv:草莓Fragaria vesca"

表3
VvARF18基因启动子顺式作用元件"
| 元件类型 Component type | 相关元件 Related component | 数量 Number | 功能注释 Functional comment |
|---|---|---|---|
| 光响应元件 Photoresponsive element | 3-AF1 binding site | 1 | 光响应元件Photoresponsive element |
| AE-box | 1 | 光反应元件Photoresponsive element | |
| Box 4 | 7 | 光响应元件Photoresponsive element | |
| G-box | 2 | 光响应元件Photoresponsive element | |
| GT1-motif | 6 | 光响应元件Photoresponsive element | |
| 激素响应元件 Hormone response element | ABRE | 2 | 脱落酸响应元件Abscisic acid response element |
| AuxRR-core | 1 | 生长素响应元件Auxin response element | |
| CGTCA-motif | 1 | 茉莉酸甲酯响应元件Methyl jasmonate response element | |
| TGACG-motif | 1 | 茉莉酸甲酯响应元件Methyl jasmonate response element | |
| P-box | 1 | 赤霉素响应元件Gibberellin response element | |
| 胁迫相关元件 Stress related component | ARE | 3 | 厌氧诱导元件Anaerobic inducing element |
| LRT | 1 | 低温诱导元件Low temperature inducing element | |
| 结合位点 Bonding component | CCAAT-box | 1 | MYBHv1结合位点MYBHv1 binding site |
| MBS | 2 | MYB参与干旱诱导位点MYB participates in drought induction sites |
| [1] |
PARK M Y, WU G, GONZALEZ-SULSER A, VAUCHERET H, POETHIG R S . Nuclear processing and export of microRNAs in Arabidopsis. Proceedings of the National Academy of Sciences of the United States of America, 2005,102(10):3691-3696.
doi: 10.1073/pnas.0405570102 pmid: 15738428 |
| [2] |
BRODERSEN P, SAKVARELIDZE-ACHARD L, BRUUN- RASMUSSEN M, DUNOYER P, YAMAMOTO Y Y, SISBURTH L, VOINNET O . Widespread translational inhibition by plant miRNAs and siRNAs. Science, 2008,320(5880):1185-1190.
doi: 10.1126/science.1159151 pmid: 18483398 |
| [3] | BUSHATI N, COHEN N . microRNA functions. Annual Review of Cell and Developmental Biology, 2007,23(1):175-205. |
| [4] |
GOSWAMI K, TRIPATHI A, SANAN-MISHRA N . Comparative miRomics of salt-tolerant and salt-sensitive rice. Journal of Integrative Bioinformatics, 2017,14(1):189-197.
doi: 10.1515/jib-2017-0002 pmid: 28637931 |
| [5] |
NAG A, JACK T . Sculpting the flower; the role of microRNAs in flower development. Current Topics in Developmental Biology, 2010,91:349-378.
doi: 10.1016/S0070-2153(10)91012-0 pmid: 20705188 |
| [6] |
CHEN Q S, LI M, ZHANG Z C, TIE W W, CHEN X, JIN L F, ZHAI N, ZHENG Q X, ZHANG J F, WANG R, XU G Y, ZHANG H, LIU P P, ZHOU H N . Integrated mRNA and microRNA analysis identifies genes and small miRNA molecules associated with transcriptional and post-transcriptional-level responses to both drought stress and re-watering treatment in tobacco. BMC Genomics, 2017,18(1):62.
doi: 10.1186/s12864-016-3372-0 pmid: 28068898 |
| [7] |
TURNER M, NIZAMPATNAM N R, BARON M, COPPIN S, DAMODARAN S, ADHIKARI S, ARUNACHALAM S P, YU O, SUBRAMANIAN S . Ectopic expression of miR160 results in auxin hypersensitivity, cytokinin hyposensitivity, and inhibition of symbiotic nodule development in soybean. Plant Physiology, 2013,162(4):2042-2055.
doi: 10.1104/pp.113.220699 |
| [8] |
PINWEHA N, ASVARAK T, VIBOONJUN U, NARANGAJAVANA J . Involvement of miR160/miR393 and their targets in cassava responses to anthracnose disease. Journal of Plant Physiology, 2015,174(1):26-35.
doi: 10.1016/j.jplph.2014.09.006 pmid: 25462963 |
| [9] |
LIU X D, HUANG J, WANG Y, KHANNA K, XIE Z X, OWEN H A, ZHAO D Z . The role of floral organs in carpels, an Arabidopsis loss-of-function mutation in MicroRNA160a, in organogenesis and the mechanism regulating its expression. Plant Journal, 2010,62(3):416-428.
doi: 10.1111/j.1365-313X.2010.04164.x pmid: 20136729 |
| [10] |
WÓJCIK A M, NODINE M D, GAJ M D . MiR160 and miR166/165 contribute to the LEC2-mediated auxin response involved in the somatic embryogenesis induction in Arabidopsis. Frontiers in Plant Science, 2017,8:2024.
doi: 10.3389/fpls.2017.02024 pmid: 29321785 |
| [11] |
MALLORY A C, BARTEL D P, BARTEL B . MicroRNA-directed regulation of Arabidopsis AUXIN RESPONSE FACTOR17 is essential for proper development and modulates expression of early auxin response genes. The Plant Cell, 2005,17(5):1360-1375.
doi: 10.1105/tpc.105.031716 pmid: 15829600 |
| [12] |
TIWARI S B, HAGEN G, GUILFOYLE T . The roles of auxin response factor domains in auxin-responsive transcription. The Plant Cell, 2003,15(2):533-543.
doi: 10.1105/tpc.008417 pmid: 12566590 |
| [13] |
GRAY W M, KEPINSKI S, ROUSE D, LEYSER O, ESTELLE M . Auxin regulates SCF TIR1-dependent degradation of Aux/IAA proteins . Nature, 2001,414(6861):271-276.
doi: 10.1038/35104500 pmid: 11713520 |
| [14] | 李艳林, 高志红, 宋娟, 王万许, 侍婷 . 植物生长素响应因子ARF与生长发育. 植物生理学报, 2017,53(10):1842-1858. |
| LI Y L, GAO Z H, SONG J, WANG W X, SHI T . Auxin response factor (ARF) and its functions in plant growth and development. Plant Physiology Journal, 2017,53(10):1842-1858. (in Chinese) | |
| [15] |
HENDELMAN A, BUXDORF K, STAV R, KRAVCHIK M, ARAZI T . Inhibition of lamina outgrowth following Solanum lycopersicum AUXIN RESPONSE FACTOR 10 (SlARF10) derepression. Plant Molecular Biology, 2012,78(6):561-576.
doi: 10.1007/s11103-012-9883-4 |
| [16] |
LIU P P, MONTGOMERY T A, FAHLGREN N, KASSCHAU K D, NONOGAKI H, CARRINGTON J C . Repression of AUXIN RESPONSE FACTOR10 by microRNA160 is critical for seed germination and post-germination stages. The Plant Journal, 2007,52(1):133-146.
doi: 10.1111/j.1365-313X.2007.03218.x pmid: 17672844 |
| [17] |
DE JONG M, WOLTERS-ARTS M, GARCIA-MARTINEZ J L, MARIANI C, VRIEZEN W H . The Solanum lycopersicum AUXIN RESPONSE FACTOR 7 (SlARF7) mediates cross-talk between auxin and gibberellin signalling during tomato fruit set and development. Journal of Experimental Botany, 2011,62(2):617-626.
doi: 10.1093/jxb/erq293 pmid: 20937732 |
| [18] |
FRIGERIO M, ALABADÍ D, PÉREZ-GÓMEZ J, GARCÍA- CÁRCEL L, PHILLIPS A L, HEDDEN P, BLÁZQUEZ M A . Transcriptional regulation of gibberellin metabolism genes by auxin signaling in Arabidopsis. Plant Physiology, 2006,142(2):553-563.
doi: 10.1104/pp.106.084871 pmid: 16905669 |
| [19] |
DORCEY E, URBEZ C, BLÁZQUEZ M A, CARBONELL J, PEREZ-AMADOR M A . Fertilization-dependent auxin response in ovules triggers fruit development through the modulation of gibberellin metabolism in Arabidopsis. The Plant Journal, 2009,58(2):318-332.
doi: 10.1111/j.1365-313X.2008.03781.x pmid: 19207215 |
| [20] |
WANG C, WANG X C, KIBET N K, SONG C N, ZHANG C Q, LI X Y, HAN J, FANG J G . Deep sequencing of grape flower and berry short RNA libraries for the discovery of new microRNAs and verification of the precise sequence of grape microRNAs preserved in miRBase. Physiologia Plantarum, 2011,143(1):64-81.
doi: 10.1111/j.1399-3054.2011.01481.x pmid: 21496033 |
| [21] |
ZHANG W Y, ABDELRAHMAN M, JIU S T, GUAN L, HAN J, ZHENG T, JIA H F, SONG C N, FANG J G, WANG C . VvmiR160s/ VvARFs, interaction and their spatio-temporal expression/cleavage products during GA-induced grape parthenocarpy. BMC Plant Biology, 2019,19(1):111.
doi: 10.1186/s12870-019-1719-9 pmid: 30898085 |
| [22] | ABU-ZAHRA T R . Percentage of thompson seeds affected by GIBBERELLIC acid and cance GIRDLING. Pakistan Journal of Botany, 2010,42(3):1755-1760. |
| [23] |
CHENG C X, XU X Z, SINGER S D, LI J, ZHANG H J, GAO M, WANG L, SONG J Y, WANG X P . Effect of GA3 treatment on seed development and seed-related gene expression in grape. PLoS ONE, 2013,8(11):e80044.
doi: 10.1371/journal.pone.0080044 pmid: 24224035 |
| [24] |
SPANUDAKIS E, JACKSON S . The role of microRNAs in the control of flowering time. Journal of Experimental Botany, 2014,65(2):365-380.
doi: 10.1093/jxb/ert453 pmid: 24474808 |
| [25] |
LUO Y, GUO Z H, LI L . Evolutionary conservation of microRNA regulatory programs in plant flower development. Developmental Biology, 2013,380(2):133-144.
doi: 10.1016/j.ydbio.2013.05.009 pmid: 23707900 |
| [26] |
ACHARD P, HERR A, BAULCOMBE D C, HARBERD N P . Modulation of floral development by a gibberellin-regulated microRNA. Development, 2004,131(14):3357-3365.
doi: 10.1242/dev.01206 pmid: 15226253 |
| [27] | 王文然, 王晨, 解振强, 贾海锋, 汤崴, 崔梦杰, 房经贵 . VvmiR397a及其靶基因VvLACs在葡萄果实发育中的作用分析. 园艺学报, 2018,45(8):1441-1455. |
| WANG W R, WANG C, XIE Z Q, JIA H F, TANG W, CUI M J, FANG J G . Function analysis of VvmiR397a and its target gene VvLACs in grape berry development. Acta Horticulturae Sinica, 2018,45(8):1441-1455. (in Chinese) | |
| [28] |
CUI M J, WANG C, ZHANG W Y, PERVAIZ T, HAIDER M S, TANG W, FANG J G . Characterization of Vv-miR156: Vv-SPL pairs involved in the modulation of grape berry development and ripening. Molecular Genetics and Genomics, 2018,293(6):1333-1354.
doi: 10.1007/s00438-018-1462-1 pmid: 29943289 |
| [29] |
YE K Y, CHEN Y, HU X W, GUO J C . Computational identification of microRNAs and their targets in apple. Genes and Genomics, 2013,35(3):377-385.
doi: 10.1111/j.1399-3054.2010.01411.x pmid: 20875055 |
| [30] |
SONG C N, FANG J G, LI X Y, LIU H, CHAO C T . Identification and characterization of 27 conserved microRNAs in citrus. Planta, 2009,230(4):671-685.
doi: 10.1007/s00425-009-0971-x pmid: 19585144 |
| [31] |
XU X B, YIN L L, YING Q C, SONG H M, XUE D W, LAI T F, XU M J, SHEN B, WANG H Z, SHI X Q . High-throughput sequencing and degradome analysis identify miRNAs and their targets involved in fruit senescence of Fragaria ananassa. PLoS ONE, 2013,8(8):e70959.
doi: 10.1371/journal.pone.0070959 pmid: 23990918 |
| [32] |
HAN J, FANG J G, WANG C, YIN Y L, SUN X, LENG X P, SONG C N . Grapevine microRNAs responsive to exogenous gibberellin. BMC Genomics, 2014,15(1):111.
doi: 10.1186/1471-2164-15-111 |
| [33] |
WANG B J, WANG J, WANG C, SHEN W B, JIA H F, ZHU X D, LI X P . Study on modes of expression and cleavage role of miR156b/c/d and its target gene Vv-SPL9 during the whole growth stage of grapevine. Journal of Heredity, 2016,107(7):626-634.
doi: 10.1093/jhered/esw030 pmid: 27660497 |
| [34] |
WANG M, WU H J, FANG J, CHU C C, WANG X J . A long noncoding RNA involved in rice reproductive development by negatively regulating osa-miR160. Science Bulletin, 2017,62(7):470-475.
doi: 10.1016/j.scib.2017.03.013 |
| [35] |
NIU J, WANG J, AN J Y, LIU L L, LIN Z X, WANG R, WANG L B, MA C, SHI L L, LIN S Z . Integrated mRNA and miRNA transcriptome reveal a cross-talk between developing response and hormone signaling for the seed kernels of Siberian apricot. Scientific Reports, 2016,6:35675.
doi: 10.1038/srep35675 pmid: 27762296 |
| [36] | CUI J, SUN Z Y, LI J L, CHENG D Y, LUO C F, DAI C H . Characterization of miRNA160/164 and their targets expression of beet (Beta vulgaris) seedlings under the salt tolerance. Plant Molecular Biology Reporter, 2018,36(5/6):790-799. |
| [37] |
LIU X D, HUANG J, WANG Y, KHANNA K, XIE Z X, OWEN H A, ZHAO D Z . The role of floral organs in carpels, an Arabidopsis loss-of-function mutation in MicroRNA160a, in organogenesis and the mechanism regulating its expression. The Plant Journal, 2010,62(3):416-428.
doi: 10.1111/j.1365-313X.2010.04164.x pmid: 20136729 |
| [38] |
DAMODHARAN S, ZHAO D Z, ARAZI T . A common miRNA160-based mechanism regulates ovary patterning, floral organ abscission and lamina outgrowth in tomato. The Plant Journal, 2016,86(6):458-471.
doi: 10.1111/tpj.13127 pmid: 26800988 |
| [1] | 李元晶, 袁瑞祥, 李永泰, 孙天歌, 刘峰, 李艳军, 张新宇. 棉花黄萎病菌β-葡萄糖苷酶基因的鉴定及其在致病中的功能[J]. 中国农业科学, 2026, 59(7): 1380-1399. |
| [2] | 张东梅, 周鑫鑫, 肖桂林, 曾祥国, 王春燕, 王泽先, 韩永超. 草莓花器应答灰霉病菌侵染的表现特征及抗病性评价方法[J]. 中国农业科学, 2026, 59(7): 1456-1466. |
| [3] | 朱嘉伟, 关璇, 饶博涵, 刘秀海, 范国元, 武运, 陶永胜. 短乳杆菌LB-21介导的酒精-苹乳三菌共发酵对干红葡萄酒色泽的影响[J]. 中国农业科学, 2026, 59(5): 1101-1110. |
| [4] | 马桂兰, 张旭阳, 李武. 鸟苷酸结合蛋白2在金黄色葡萄球菌诱导巨噬细胞凋亡中的调控作用[J]. 中国农业科学, 2026, 59(4): 912-926. |
| [5] | 张梦博, 谭鸿冰, 沈甜, 徐美隆, 周新明, 房玉林, 鞠延仑. 不同灌溉量与抗蒸腾剂处理对葡萄酒品质的影响[J]. 中国农业科学, 2026, 59(2): 413-426. |
| [6] | 冯伟晴, 倪媛蒨, 费腾, 李有梅, 谢兆森. 不同果形葡萄果实维管束形态结构、分布特征及其水分运输功能差异[J]. 中国农业科学, 2026, 59(1): 161-178. |
| [7] | 王思琪, 邹利人, 白瑞雯, 闫可, 王思洋, 齐晓光, 申海林, 温景辉. 赤霉素调控‘蜜汁’葡萄穗轴硬化关键基因的挖掘[J]. 中国农业科学, 2026, 59(1): 179-189. |
| [8] | 谭西北, 兰徐颖, 刘崇怀, 樊秀彩, 姜建福, 孙磊, 李鹏, 余书鑫, 张颖. 不同抗性葡萄响应白腐病侵染的次生代谢物变化[J]. 中国农业科学, 2025, 58(9): 1767-1778. |
| [9] | 陈龙云, 胡俊强, 何灿, 史建荣, 徐剑宏, 王刚. 利用大孔吸附树脂与高速逆流色谱纯化脱氧雪腐镰刀菌烯醇-3-葡萄糖苷[J]. 中国农业科学, 2025, 58(8): 1627-1637. |
| [10] | 汤学燊, 党仕卓, 周娟, 李佳豪, 李梅花, 胡豪, 张亚红. 基于红蓝光调控分析VvBES1-1参与‘红地球’葡萄花芽的分化[J]. 中国农业科学, 2025, 58(8): 1650-1662. |
| [11] | 杨彩丽, 李永洲, 贺亮亮, 宋银花, 章鹏, 刘肇先, 李鹏慧, 刘三军. 葡萄TPS基因家族全基因组鉴定及VvTPS4在单萜形成中的功能验证[J]. 中国农业科学, 2025, 58(7): 1397-1417. |
| [12] | 张天雨, 李白, 藏金萍, 曹宏哲, 董金皋, 邢继红, 张康. 灰葡萄孢HMG家族基因的全基因组鉴定与表达规律分析[J]. 中国农业科学, 2025, 58(4): 704-718. |
| [13] | 郭奥琳, 林俊璇, 赖恭梯, 贺丽媛, 车建美, 潘若, 杨方学, 黄玉吉, 陈桂信, 赖呈纯. 过表达VdF3′5′H2对刺葡萄细胞花青素组分积累的影响[J]. 中国农业科学, 2025, 58(4): 802-818. |
| [14] | 从琪琪, 张静怡, 孟祥龙, 戴蓬博, 李波, 胡同乐, 王树桐, 曹克强, 王亚南. 我国苹果轮纹病弱毒菌株中病毒的鉴定及其携带情况的检测[J]. 中国农业科学, 2025, 58(3): 478-492. |
| [15] | 张向昆, 李佳莹, 乔如梦, 何静蕾, 王莉, 师校欣, 杜国强. 不同锌水平下GFabV对‘阳光玫瑰’葡萄光合效率及光合相关基因表达的影响[J]. 中国农业科学, 2025, 58(24): 5190-5200. |
|
||