













中国农业科学 ›› 2019, Vol. 52 ›› Issue (14): 2436-2449.doi: 10.3864/j.issn.0578-1752.2019.14.005
收稿日期:2019-03-22
接受日期:2019-04-24
出版日期:2019-07-16
发布日期:2019-07-26
通讯作者:
王跃进
作者简介:刘梦琦,E-mail: mengqil@163.com。
基金资助:
LIU MengQi,WU FengYing,WANG YueJin( )
)
Received:2019-03-22
Accepted:2019-04-24
Online:2019-07-16
Published:2019-07-26
Contact:
YueJin WANG
摘要:
【目的】葡萄是世界性重要果树,欧洲葡萄品种因其优质高产被广泛栽培,但其突出缺点是抗病性弱,尤其易受白粉病危害。葡萄中芪合成酶(stilbene synthase,STS)的代谢产物白藜芦醇是植物体内具有抗病功能的植保素,果实中的白藜芦醇对人具有保健作用。中国野生毛葡萄‘丹凤-2’抗病性强且白藜芦醇含量高。论文旨在研究中国野生毛葡萄‘丹凤-2’芪合成酶基因(STS)及其功能,应用于抗病育种来提高欧洲葡萄抗病性及果实中芪类物质含量。【方法】同源克隆中国野生毛葡萄‘丹凤-2’芪合成酶基因VqSTS26和VqSTS32,构建pCAMBIA35S::VqSTSs::GFP过表达载体;以器官发生途径诱导的无核白分生愈伤组织作为受体材料,采用农杆菌介导法进行遗传转化,获得转基因葡萄植株;分别从转录水平和代谢产物水平比较转基因与野生型无核白在自然生长条件与人工接种葡萄白粉病菌(Uncinula necator)诱导下STS表达及芪类物质产生与积累的差异;通过显微观察白粉病菌在转基因与野生型无核白叶片上的生长发育进程,统计孢子萌发、菌丝生长与分生孢子梗形成数目,对转基因植株进行抗病性分析。【结果】通过PCR检测和Western blot鉴定,获得了稳定转化VqSTS26植株8株和稳定转化VqSTS32植株5株。在自然生长条件下实时荧光定量PCR分析表明VqSTS26、VqSTS32转基因无核白STS的表达量显著提高,芪合成酶上游基因PAL与下游基因RSGT的表达量上调,而与芪合成酶存在底物竞争关系的CHS表达下调;液相色谱分析表明芪类物质主要以糖苷的反式云杉新苷形式存在,VqSTS26、VqSTS32转基因无核白芪类物质的含量极显著高于野生型无核白。STS的表达及其产物合成受白粉病菌诱导,随白粉病菌诱导,STS表达量在1—2 dpi时显著升高,至7 dpi时表达量达最高;诱导表达产生的芪类物质由原来的反式云杉新苷新增加了反式白藜芦醇和葡萄素,且含量增加;转基因植株在STS表达量、芪类物质的积累方面均极显著高于野生型无核白。显微观察白粉病菌在葡萄叶片上的生长发育状态,对比野生型无核白,转基因植株白粉病菌生长受到抑制,菌丝发育更迟,7 dpi时转基因葡萄叶片上的分生孢子梗数量低于野生型无核白。【结论】过表达中国野生毛葡萄‘丹凤-2’VqSTS26和VqSTS32可以提高无核白中STS的表达量,促进芪类物质的形成与积累,抑制转基因无核白叶片上白粉病菌的生长。因此,中国野生毛葡萄‘丹凤-2’与其携带的STS及其产物,是定向改良欧洲葡萄品种白粉病抗性与芪类物质含量的重要种质资源与基因资源。
刘梦琦,吴凤颖,王跃进. 中国野生毛葡萄芪合成酶基因表达与抗白粉病分析[J]. 中国农业科学, 2019, 52(14): 2436-2449.
LIU MengQi,WU FengYing,WANG YueJin. Expression of Stilbene Synthase Gene and Resistance to Powdery Mildew Analysis of Chinese Wild Vitis quinquangularis[J]. Scientia Agricultura Sinica, 2019, 52(14): 2436-2449.
表1
本研究所用引物"
| 基因 Gene | 引物序列 Primer sequence | 目标大小 Target size (bp) |
|---|---|---|
| VqSTS26 (Vector construction, PCR detection) | F: AGAACACGGGGGACGAGCTCATGGCTTCAGTTGAGGAATTTAGAAACG R: ACCATGGTGTCGACTCTAGAATTTGTAACTGTAGGAATGCTATGCAGC | 1179 |
| VqSTS32 (Vector construction, PCR detection) | F: AGAACACGGGGGACGAGCTCATGGCTTCAATTGAGGAAATTAGAAACGC R: ACCATGGTGTCGACTCTAGAATTTGTAACCATAGGAACGCTATGCAG | 1179 |
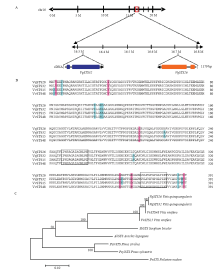
图1
‘丹凤-2’VqSTS26、VqSTS32的定位、序列比对及聚类分析 A:VqSTS26、VqSTS32的染色体定位分析Chromosome localization analysis of VqSTS26, VqSTS32;B:VqSTS26、VqSTS32与欧洲葡萄同源蛋白氨基酸序列比对;阴影部分为差异位点,方框部分为保守序列Amino acid sequence alignment of homologous proteins between VqSTS26, VqSTS32 and STS genes from V. vinifera, the shading is the difference site and the box part is the conservative sequence;C:VqSTS26、VqSTS32与欧洲葡萄、高粱、花生、白松、樟子松和松叶兰芪合成酶基因氨基酸序列聚类分析,VqSTS26(AFM56643.1)、VqSTS32(AFM56649.1)、VvSTS15(XP_002268756.1)、VvSTS48(NP_001267934.1)、SbSTS(AAL49965)、AhSTS(BAA78617)、PstrSTS(CAA87012)、PsylSTS(CAA43165)、PnSTS(BAA87924)Cluster analysis of STS amino acid sequence from VqSTS26, VqSTS32 and V. vinifera, Sorghum bicolor, Arachis hypogaea, Pinus strobus, Pinus sylvestris, Psilotum nudum"
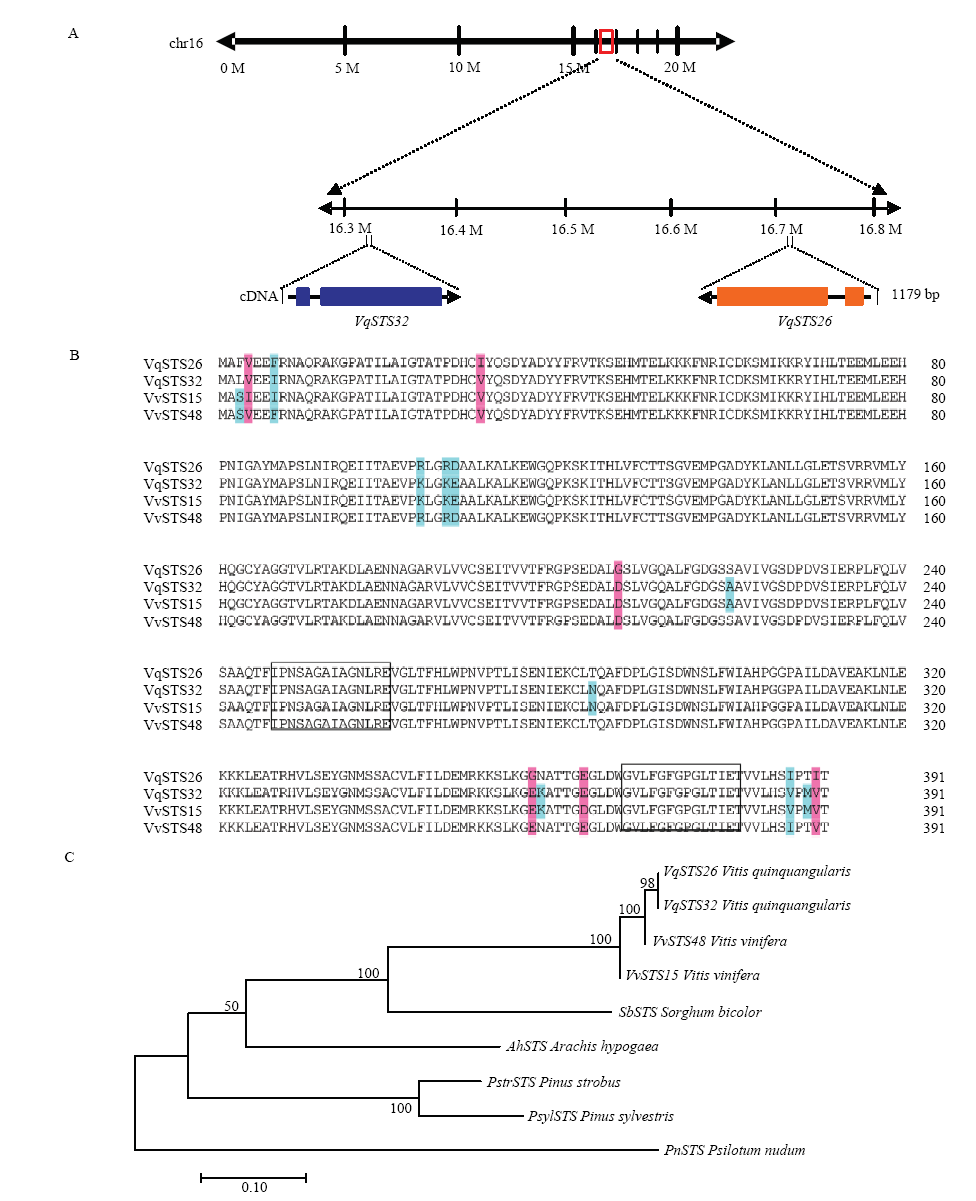

图2
转VqSTS26、VqSTS32无核白的遗传转化过程与转基因植株的鉴定 A:基因克隆与过表达载体构建Gene cloning and vector construction。a:pCAMBIA35S:: VqSTSs:: GFP载体 pCAMBIA35S:: VqSTSs:: GFP vector;b:VqSTS26、VqSTS32目的基因扩增Gene amplification of VqSTS26 and VqSTS32;c:VqSTS26、VqSTS32基因连接表达载体pCAMBIA2300的双酶切检测,CK为pCAMBIA2300的空载体对照 Double enzyme digestion detection of recombinant plasmid, CK was the empty vector as control;d:VqSTS26、VqSTS32转化农杆菌GV3101的PCR检测,CK1、CK2分别为每个基因重组质粒的阳性对照 PCR detection of agrobacterium GV3101 transformed with VqSTS26 and VqSTS32. CK1 and CK2 were positive controls of recombinant plasmid for each gene。M:Trans 2k plus DNA maker。B:无核白分生愈伤组织的诱导(a—d)、遗传转化(e—h)与转化后的诱导成苗、扩繁与移栽炼苗(i—n) Induction (a-d) and genetic transformation (e-h) of Thompson Seedless meristem callus and induction into seedlings after transformation (i-n)。C:无核白葡萄转VqSTS26、VqSTS32抗性株系的PCR检测和Western blot检测PCR amplification and Western blot detection of Thompson Seedless transgenic lines with VqSTS26 and VqSTS32"

表2
葡萄叶片每100个孢子萌发数、初级菌丝数、次级菌丝数和分生孢子梗数统计"
| 株系 Line | 1 dpi | 2 dpi | 3 dpi | 7 dpi | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 萌发数 Germination | 初级菌 丝数 Primary hyphae | 次级菌 丝数 Secondary hyphae | 萌发数 Germination | 初级菌 丝数 Primary hyphae | 次级菌 丝数 Secondary hyphae | 萌发数 Germination | 初级菌 丝数 Primary hyphae | 次级菌丝数 Secondary hyphae | 分生孢子梗数 Conidiophore | 分生孢子 梗数 Conidiophore | |
| 野生型 Wide type | 42.67± 3.51a | 21.67± 2.08a | 4.33± 1.53a | 22.67± 2.08a | 8.67± 1.15c | 50.33± 3.51a | 6.33± 1.53c | 12.67± 1.53c | 69.00± 4.00a | 15.33±1.53a | 195.33± 12.66a |
| OEVqSTS26-L6 | 32.33± 2.52bc | 7.33± 1.53b | 0.33± 0.58b | 16.33± 0.58b | 11.67± 1.53c | 14.33± 2.08c | 30.67± 1.53a | 13.33± 1.53c | 16.33± 2.52c | 0 | 92.67± 7.37b |
| OEVqSTS26-L8 | 26.00± 1.00c | 12.00± 2.00b | 0.33± 0.58b | 17.33± 1.53b | 17.67± 2.52b | 10.33± 1.53c | 31.67± 3.51a | 11.00± 1.73c | 23.00± 2.65c | 0 | 42.00± 3.00c |
| OEVqSTS32-L3 | 35.00± 1.53b | 17.67± 2.52a | 3.33± 1.53ab | 17.00± 1.00b | 25.00± 1.00a | 22.67± 1.53b | 13.33± 2.52b | 18.00± 1.00b | 34.33± 3.06b | 0 | 110.33± 14.29b |
| OEVqSTS32-L4 | 35.33± 1.53b | 19.67± 1.53a | 3.67± 1.53a | 13.67± 1.53b | 21.00± 2.00ab | 26.33± 1.53b | 18.33± 1.53b | 23.00± 2.00a | 32.33± 2.08b | 0 | 58.33± 2.08c |
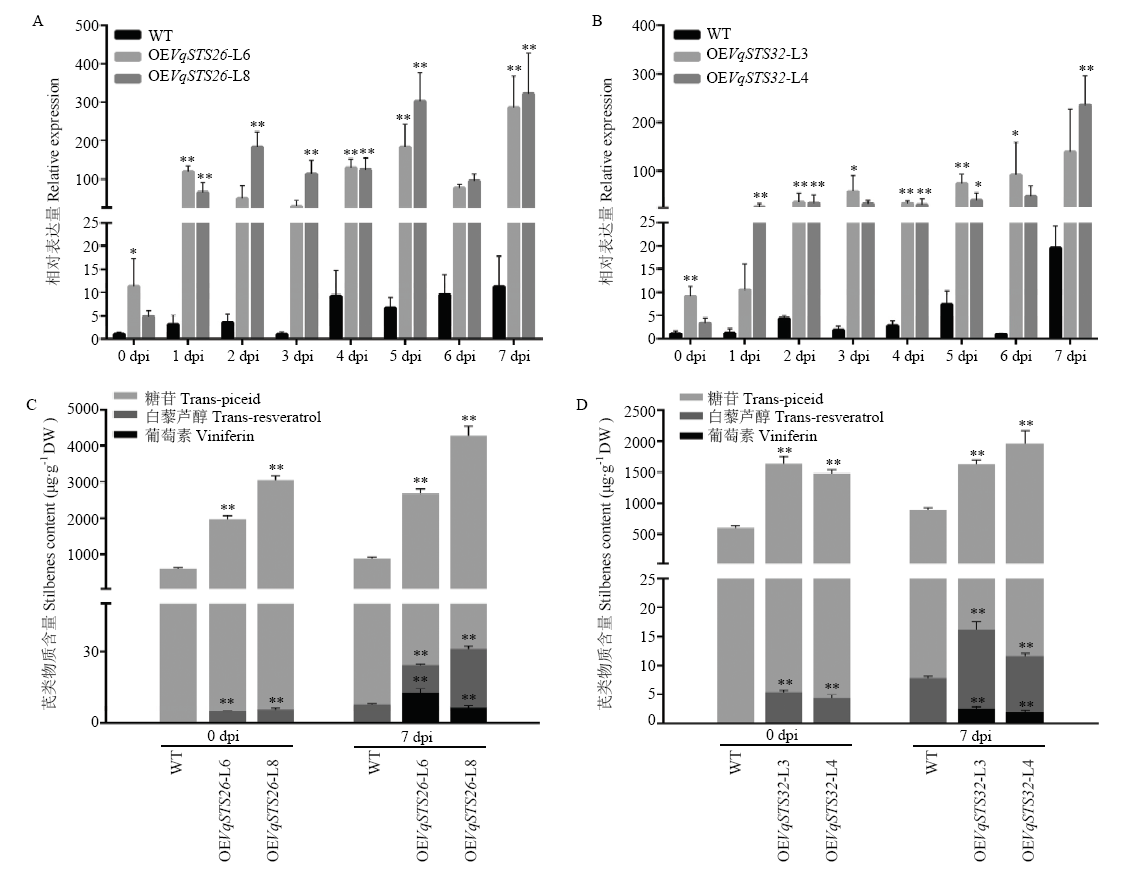
图4
人工接种白粉病菌后转基因植株与野生型无核白STS表达及芪类物质含量分析 A、B:qRT-PCR分析VqSTS26(A)和VqSTS32(B)转基因与野生型无核白接菌0—7 dpi叶片STS的表达量 qRT-PCR analysis of STS expression between transgenic grapes with overexpression VqSTS26 (A) and VqSTS32 (B) and wild-type Thompson Seedless under U. necator induction at 0-7 dpi;C、D:HPLC分析VqSTS26(C)和VqSTS32(D)转基因与野生型无核白接菌前后芪类物质含量的变化HPLC detection of stilbenoids content in transgenic grapes with overexpression VqSTS26 (C) and VqSTS32 (D) and wild-type Thompson Seedless under U. necator induction at 0 and 7 dpi. *P<0.05,**P<0.01"
| [1] |
ALLEWELDT G, POSSINGHAM J V . Progress in grapevine breeding. Theoretical and Applied Genetics, 1988,75(5):669-673.
doi: 10.1007/BF00265585 |
| [2] | 贺普超 . 葡萄学. 北京:中国农业出版社, 1999. |
| HE P C. Viticulture. Beijing: China Agriculture Press, 1999. ( in Chinese) | |
| [3] | 贺普超, 王跃进, 王国英, 任志邦, 和纯成 . 中国葡萄属野生种抗病性的研究. 中国农业科学, 1991,24(3):50-56. |
| HE P C, WANG Y J, WANG G Y, REN Z B, HE C C . The studies on the disease-resistance of Vitis wild species originated in China. Scientia Agricultura Sinica, 1991,24(3):50-56. (in Chinese) | |
| [4] | GOMÈS E, COUTOS-THÉVENOT P . Molecular aspects of grapevine- pathogenic fungi interactions//ROUBELAKIS-ANGELAKIS K A. Grapevine Molecular Physiology and Biotechnology. 2nd ed. Springer Science+Business Media B.V., 2009: 407-428. |
| [5] | RIAZ S, DOLIGEZ A, HENRY R J, WALKER M A, KOLE C . Grape. Fruits and Nuts, 2007,39(4):63-101. |
| [6] |
BISSON L F, WATERHOUSE A L, EBELER S E, WALKER M A, LAPSLEY J T . The present and future of the international wine industry. Nature, 2002,418(6898):696-699.
doi: 10.1038/nature01018 |
| [7] | WANG Y, LIU Y, HE P, CHEN J, LAMIKANRA O, LU J . Evaluation of foliar resistance to Uncinula necator in Chinese wild Vitis species. Vitis, 1995,4:131-138. |
| [8] |
ZHOU Q, DU Y, CHENG S, LI R, ZHANG J, WANG Y . Resveratrol derivatives in four tissues of six wild Chinese grapevine species. New Zealand Journal of Crop and Horticultural Science, 2015,43(3):204-213.
doi: 10.1080/01140671.2015.1010547 |
| [9] |
SCHNEE S, VIRET O, GINDRO K . Role of stilbenes in the resistance of grapevine to powdery mildew. Physiological and Molecular Plant Pathology, 2008,72:128-133.
doi: 10.1016/j.pmpp.2008.07.002 |
| [10] |
SMOLIGA J M, BAUR J A, HAUSENBLAS H A . Resveratrol and health—A comprehensive review of human clinical trials. Molecular Nutrition and Food Research, 2011,55(8):1129-1141.
doi: 10.1002/mnfr.v55.8 |
| [11] |
JANG M, CAI L, UDEANI G O, SLOWING K V, THOMAS C F, BEECHER C W W, FONG H H S, FARNSWORTH N R, KINGHORN A D, MEHTA R G, MOON R C, PEZZUTO J M . Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science, 1997,275(5297):218-220.
doi: 10.1126/science.275.5297.218 |
| [12] | RUPPRICH N, KINDL H . Stilbene synthases and stilbenecarboxylate synthases, I Enzymatic synthesis of 3,5,4-trihydroxystilbene from p-coumaroyl coenzyme A and malonyl coenzyme A. Hoppe-Seyler's Zeitschrift für Physiologische Chemie, 1978,359(2):165-172. |
| [13] | LISWIDOWATI, MELCHIOR F, HOHMANN F, SCHWER B, KINDL H . Induction of stilbene synthase by Botrytis cinerea in cultured grapevine cells. Planta, 1991,183(2):307-314. |
| [14] |
HART J H . Role of phytostilbenes in decay and disease resistance. Annual Review of Phytopathology, 1981,19:437-458.
doi: 10.1146/annurev.py.19.090181.002253 |
| [15] |
PARAGE C, TAVARES R, RÉTY S, BALTENWECK-GUYOT R, POUTARAUD A, RENAULT L, HEINTZ D, LUGAN R, MARAIS G, AUBOURG S, HUGUENEY P . Structural, functional, and evolutionary analysis of the unusually large stilbene synthase gene family in grapevine. Plant Physiology, 2012,160(3):1407-1419.
doi: 10.1104/pp.112.202705 |
| [16] | HAIN R, BIESELER B, KINDL H, SCHRÖDER G, STÖCKER R . Expression of a stilbene synthase gene in Nicotiana tabacum results in synthesis of the phytoalexin resveratrol. Plant Molecular Biology, 1990,15(2):325-335. |
| [17] |
HAIN R, REIF H J, KRAUSE E, LANGEBARTELS R, KINDL H, VORNAM B, WIESE W, SCHMELZER E, SCHREIER P H, STÖCKER R H, STENZEL K . Disease resistance results from foreign phytoalexin expression in a novel plant. Nature, 1993,361(6408):153-156.
doi: 10.1038/361153a0 |
| [18] | STARK-LORENZEN P, NELKE B, HÄNßLER G, MÜHLBACH H P, THOMZIK J E . Transfer of a grapevine stilbene synthase gene to rice ( Oryza sativa L.). Plant Cell Reports, 1997,16(10):668-673. |
| [19] | NICOLETTI I, DE ROSSI A, GIOVINAZZO G, CORRADINI D . Identification and quantification of stilbenes in fruits of transgenic tomato plants (Lycopersicon esculentum Mill.) by reversed phase HPLC with photodiode array and mass spectrometry detection. Journal of Agricultural and Food Chemistry, 2007,55(9):3304-3311. |
| [20] | YU C K, LAM C N, SPRINGOB K, SCHMIDT J, CHU I K, LO C . Constitutive accumulation of cis-piceid in transgenic Arabidopsis overexpressing a sorghum stilbene synthase gene. Plant and Cell Physiology, 2006,47(7):1017-1021. |
| [21] |
RÜHMANN S, TREUTTER D, FRITSCHE S, BRIVIBA K, SZANKOWSKI I . Piceid (resveratrol glucoside) synthesis in stilbene synthase transgenic apple fruit. Journal of Agricultural and Food Chemistry, 2006,54(13):4633-4640.
doi: 10.1021/jf060249l |
| [22] | DAI L, ZHOU Q, LI R, DU Y, HE J, WANG D, CHENG S, ZHANG J, WANG Y . Establishment of a picloram-induced somatic embryogenesis system in Vitis vinifera cv. chardonnay and genetic transformation of a stilbene synthase gene from wild-growing Vitis species. Plant Cell, Tissue and Organ Culture, 2015,121(2):397-412. |
| [23] | CHENG S, XIE X, XU Y, ZHANG C, WANG X, ZHANG J, WANG Y . Genetic transformation of a fruit-specific, highly expressed stilbene synthase gene from Chinese wild Vitis quinquangularis. Planta, 2016,243(4):1041-1053. |
| [24] |
VANNOZZI A, DRY I B, FASOLI M, ZENONI S, LUCCHIN M . Genome-wide analysis of the grapevine stilbene synthase multigenic family: Genomic organization and expression profiles upon biotic and abiotic stresses. BMC Plant Biology, 2012,12:130.
doi: 10.1186/1471-2229-12-130 |
| [25] | SHI J, HE M, CAO J, WANG H, DING J, JIAO Y, LI R, HE J, WANG D, WANG Y . The comparative analysis of the potential relationship between resveratrol and stilbene synthase gene family in the development stages of grapes (Vitis quinquangularis and Vitis vinifera). Plant Physiology and Biochemistry, 2014,74:24-32. |
| [26] |
XIE X, AGÜERO C B, WANG Y, WALKER M A . Genetic transformation of grape varieties and rootstocks via organogenesis. Plant Cell, Tissue and Organ Culture, 2016,126(3):541-552.
doi: 10.1007/s11240-016-1023-4 |
| [27] | 鲍睿 . 中国野生毛葡萄VqRSGT基因的遗传转化与功能研究[D]. 杨陵: 西北农林科技大学, 2017. |
| BAO R . A study on the genetic transformation and functional analysis of VqRSGT from Chinese wild Vitis quinquangularis[D]. Yangling: Northwest A & F University, 2017. ( in Chinese) | |
| [28] | MICALI C, GÖLLNER K, HUMPHRY M, CONSONNI C, PANSTRUGA R . The powdery mildew disease of Arabidopsis: A paradigm for the interaction between plants and biotrophic fungi//The Arabidopsis Book. The American Society of Plant Biologists, 2008,6:e0115. |
| [29] |
CHONG J, POUTARAUD A, HUGUENEY P . Metabolism and roles of stilbenes in plants. Plant Science, 2009,177(3):143-155.
doi: 10.1016/j.plantsci.2009.05.012 |
| [30] |
FERRER J L, AUSTIN M B, JR STEWART C, NOEL J P . Structure and function of enzymes involved in the biosynthesis of phenylpropanoids. Plant Physiology and Biochemistry, 2008,46(3):356-370.
doi: 10.1016/j.plaphy.2007.12.009 |
| [31] | BROWN M V, MOORE J N, FENN P, MCNEW R W . Evaluation of grape germplasm for downy mildew resistance. Fruit Varieties Journal, 1999,53(1):22-29. |
| [32] | MALACARNE G, VRHOVSEK U, ZULINI L, CESTARO A, STEFANINI M, MATTIVI F, DELLEDONNE M, VELASCO R, MOSER C . Resistance to Plasmopara viticola in a grapevine segregating population is associated with stilbenoid accumulation and with specific host transcriptional responses. BMC Plant Biology, 2011,11:114. |
| [33] |
LANGCAKE P, PRYCE R J . The production of resveratrol and the viniferins by grapevines in response to ultraviolet irradiation. Phytochemistry, 1977,16(8):1193-1196.
doi: 10.1016/S0031-9422(00)94358-9 |
| [34] | LANGCAKE P, MCCARTHY W V . The relationship between resveratrol production to infection of grapevine leaves by Botrytis cinerea. Vitis, 1979,18:244-253. |
| [35] |
ROMERO-PÉREZ A I, LAMUELA-RAVENTÓS R M, ANDRÉS- LACUEVA C, TORRE-BORONAT M C . Method for the quantitative extraction of resveratrol and piceid isomers in grape berry skins. Effect of powdery mildew on the stilbene content. Journal of Agricultural and Food Chemistry, 2001,49(1):210-215.
doi: 10.1021/jf000745o |
| [36] |
PEZET R, GINDRO K, VIRET O, SPRING J L . Glycosylation and oxidative dimerization of resveratrol are respectively associated to sensitivity and resistance of grapevine cultivars to downy mildew. Physiological and Molecular Plant Pathology, 2004,65(6):297-303.
doi: 10.1016/j.pmpp.2005.03.002 |
| [37] | NICOTRA S, CRAMAROSSA M R, MUCCI A, PAGNONI U M, RIVA S, FORTI L . Biotransformation of resveratrol: Synthesis of trans-dehydrodimers catalyzed by laccases from Myceliophtora thermophyla and from Trametes pubescens. Tetrahedron, 2004,60(3):595-600. |
| [38] | COUTOS-THÉVENOT P, POINSSOT B, BONOMELLI A, YEAN H, BREDA C, BUFFARD D, ESNAULT R, HAIN R, BOULAY M . In vitro tolerance to Botrytis cinerea of grapevine 41B rootstock in transgenic plants expressing the stilbene synthase Vst1 gene under the control of a pathogen-inducible PR10 promoter. Journal of Experimental Botany, 2001,52(358):901-910. |
| [39] |
SCHRÖDER J, SCHRÖDER G . Stilbene and chalcone synthases: Related enzymes with key functions in plant-specific pathways. Zeitschrift Für Naturforschung C, Journal of Biosciences, 1990,45(1/2):1-8.
doi: 10.1515/znc-1990-1-202 |
| [40] |
WEISSHAAR B, JENKINS G I . Phenylpropanoid biosynthesis and its regulation. Current Opinion in Plant Biology, 1998,1(3):251-257.
doi: 10.1016/S1369-5266(98)80113-1 |
| [41] | KOBAYASHI S, DING C K, NAKAMURA Y, NAKAJIMA I, MATSUMOTO R . Kiwifruits (Actinidia deliciosa) transformed with a Vitis stilbene synthase gene produce piceid (resveratrol-glucoside). Plant Cell Reports, 2000,19(9):904-910. |
| [42] | SCHWEKENDIEK A, SPRING O, HEYERICK A, PICKEL B, PITSCH NT, PESCHKE F, DE KEUKELEIRE D, WEBER G . Constitutive expression of a grapevine stilbene synthase gene in transgenic hop (Humulus lupulus L.) yields resveratrol and its derivatives in substantial quantities. Journal of Agricultural and Food Chemistry, 2007,55(17):7002-7009. |
| [43] | MULLINS M G, TANG F C A, FACCIOTTI D . Agrobacterium- mediated genetic transformation of grapevines: Transgenic plants of Vitis rupestris Scheele and buds of Vitis vinifera L. Nature Biotechnology, 1990,8(11):1041-1045. |
| [1] | 赵海霞,肖欣,董玘鑫,吴花拉,李成磊,吴琦. 苦荞愈伤遗传转化体系的优化及用于FtCHS1的过表达分析[J]. 中国农业科学, 2022, 55(9): 1723-1734. |
| [2] | 蔡苇荻,张羽,刘海燕,郑恒彪,程涛,田永超,朱艳,曹卫星,姚霞. 基于成像高光谱的小麦冠层白粉病早期监测方法[J]. 中国农业科学, 2022, 55(6): 1110-1126. |
| [3] | 郭泽西,孙大运,曲俊杰,潘凤英,刘露露,尹玲. 查尔酮合成酶基因在葡萄抗灰霉病和霜霉病中的作用[J]. 中国农业科学, 2022, 55(6): 1139-1148. |
| [4] | 冯子恒,宋莉,张少华,井宇航,段剑钊,贺利,尹飞,冯伟. 基于无人机多光谱和热红外影像信息融合的小麦白粉病监测[J]. 中国农业科学, 2022, 55(5): 890-906. |
| [5] | 胡朝月, 王凤涛, 郎晓威, 冯晶, 李俊凯, 蔺瑞明, 姚小波. 小麦抗条锈病基因对中国条锈菌主要流行小种的抗性分析[J]. 中国农业科学, 2022, 55(3): 491-502. |
| [6] | 张洁,姜长岳,王跃进. 中国野生毛葡萄转录因子VqWRKY6与VqbZIP1互作调控抗白粉病功能分析[J]. 中国农业科学, 2022, 55(23): 4626-4639. |
| [7] | 张琦,段玉,苏越,蒋琪琪,王春庆,宾羽,宋震. 基于柑橘叶斑驳病毒的表达载体构建及应用[J]. 中国农业科学, 2022, 55(22): 4398-4407. |
| [8] | 康忱,赵雪芳,李亚栋,田哲娟,王鹏,吴志明. 黄瓜CC-NBS-LRR家族基因鉴定及在霜霉病和白粉病胁迫下的表达分析[J]. 中国农业科学, 2022, 55(19): 3751-3766. |
| [9] | 储宝华,曹富国,卞宁宁,钱谦,李中兴,李雪薇,刘泽远,马锋旺,管清美. 84个苹果栽培品种对斑点落叶病的抗性评价和全基因组关联分析[J]. 中国农业科学, 2022, 55(18): 3613-3628. |
| [10] | 李依镁,王娇,王萍,师恺. 番茄糖转运蛋白SlSTP2在防御细菌性叶斑病中的功能[J]. 中国农业科学, 2022, 55(16): 3144-3154. |
| [11] | 方瀚墨,胡璋健,马巧梅,丁淑婷,王萍,王安然,师恺. 番茄SlβCA3在防御丁香假单胞菌番茄致病变种中的功能[J]. 中国农业科学, 2022, 55(14): 2740-2751. |
| [12] | 方桃红,张敏,马春花,郑晓晨,谭文静,田冉,燕琼,周新力,李鑫,杨随庄,黄可兵,王建锋,韩德俊,王晓杰,康振生. 小麦抗条锈基因Yr52在品种改良中的应用[J]. 中国农业科学, 2022, 55(11): 2077-2091. |
| [13] | 沙仁和,兰黎明,王三红,罗昌国. 苹果转录因子MdWRKY40b抗白粉病的机理[J]. 中国农业科学, 2021, 54(24): 5220-5229. |
| [14] | 丁茜,赵凯茜,王跃进. 中国野生毛葡萄芪合酶基因表达及对葡萄抗白粉病的影响[J]. 中国农业科学, 2021, 54(2): 310-323. |
| [15] | 张勇,阎俊,肖永贵,郝元峰,张艳,徐开杰,曹双河,田宇兵,李思敏,闫俊良,张赵星,陈新民,王德森,夏先春,何中虎. 中麦895高产稳产优质特性遗传解析[J]. 中国农业科学, 2021, 54(15): 3158-3167. |
|
||